A carbohydrate is a biomolecule consisting of carbon, hydrogen and oxygen atoms, usually with a carbon-hydrogen–oxygen atom ratio of 1:2:1.
Carbohydrates can be classified as monosaccharides, disaccharides, and polysaccharides based on the amount of monomer units present.
Carbohydrates can be classified as monosaccharides, disaccharides, and polysaccharides based on the amount of monomer units present.
Monosaccharide
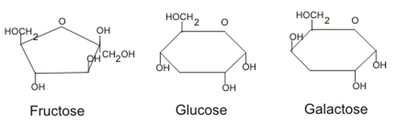
- The monomer unit for carbohydrates is a monosaccharide or a simple sugar.
- Function: Main source of energy for the cell.
- Function: Main source of energy for the cell.
Disaccharide
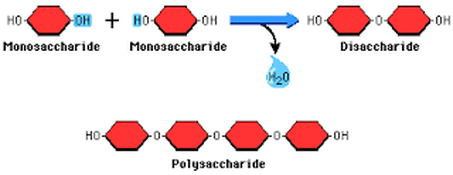
- When two of these monosaccharides are linked by covalent bonds a disaccharide is created.
- Glycosidic bond (linkage) is a type of covalent bond that joins a carbohydrate (sugar) molecule to another group
- Glycosidic bond (linkage) is a type of covalent bond that joins a carbohydrate (sugar) molecule to another group
Polysaccharide
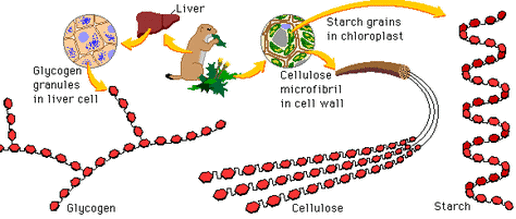
- When several monosaccharides are bonded together a polysaccharide, or complex sugar, is created.
- Polysaccharides are the polymers of carbohydrates.
- They Include:
Starch (storage of energy in plants)
Glycogen (storage of energy in animals and yeast)
Cellulose (structural support in plant cell walls)
Chitin (structural support in animal-exoskeleton of insects, and in cell walls of fungi)
- Polysaccharides are insoluble in water, do NOT taste sweet and do NOT crystalize
- Polysaccharides are the polymers of carbohydrates.
- They Include:
Starch (storage of energy in plants)
Glycogen (storage of energy in animals and yeast)
Cellulose (structural support in plant cell walls)
Chitin (structural support in animal-exoskeleton of insects, and in cell walls of fungi)
- Polysaccharides are insoluble in water, do NOT taste sweet and do NOT crystalize


