Standard 18.12 _ Water and Life
Discuss the special properties of water that contribute to Earth's suitability as an environment for life: cohesive behavior, ability to moderate temperature, expansion upon freezing, and versatility as a solvent
Practice Questions here & here
Quick Facts about water:
1. Water is the most important compound in living
things.
2. Water serves as a transport material in organisms (i.e. blood in animals and sap in plants)
3. Water makes up 75-90% of an organism.
things.
2. Water serves as a transport material in organisms (i.e. blood in animals and sap in plants)
3. Water makes up 75-90% of an organism.
Properties of Water
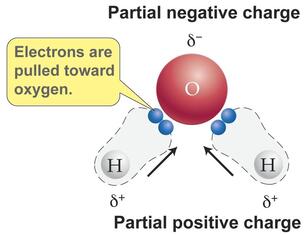
POLAR
- Molecule with an unequal distribution of charge (one end is + and the other is -)
- Attract ions as well as other polar molecules, thus it can
dissolve ions compounds such as salt and polar molecules such as sugar.
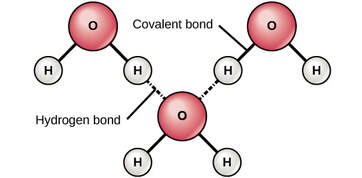
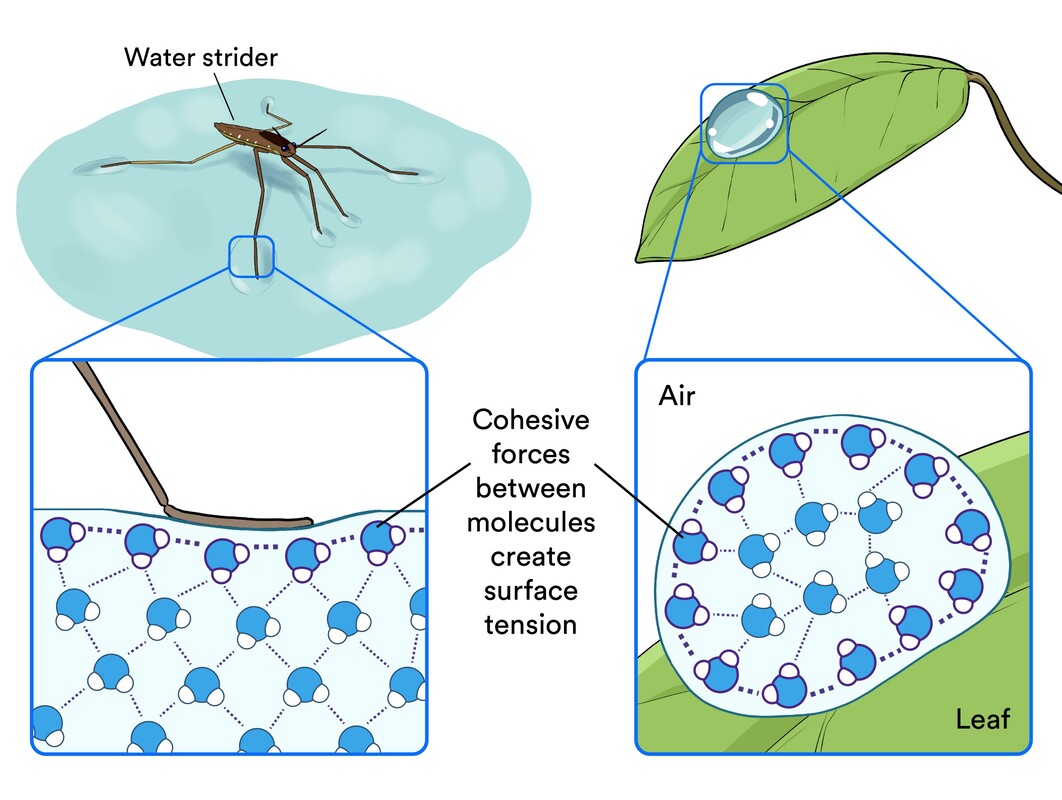
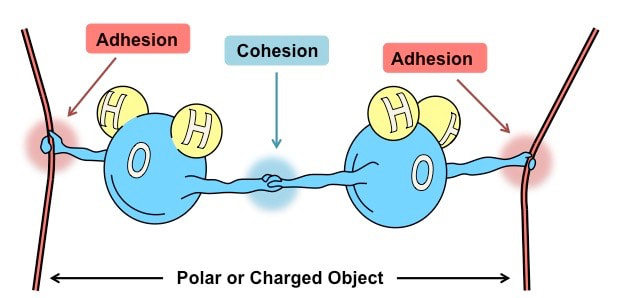
COHESION
- Water molecules attract other water molecules
- Positive charge of hydrogen and attracts the negative charge oxygen of another water molecule. This attraction is called a "hydrogen bond"
- It causes water drops and bodies of water to form.
|
SURFACE TENSION - DUE TO COHESSION
|
ADHESION
- Water sticks to other molecules
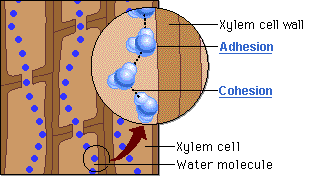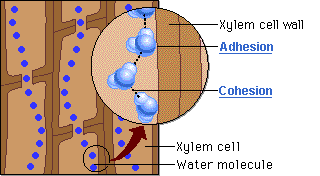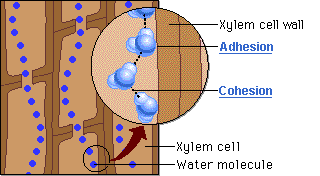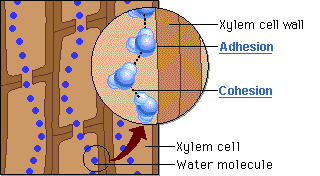
CAPILLARY ACTION
- Can move up thin tubes
- Ability of a substance to draw a liquid up against the force of gravity
- Both ADHESION and COHESION are responsible for water transport in plants.
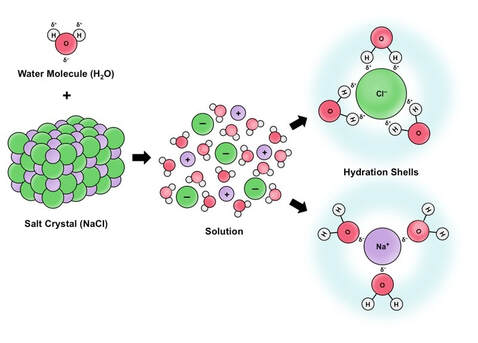
VERSATILITY AS A SOLVENT
- It's often called the “universal solvent” because water
can dissolve more substances than any other liquid. - Some substances, like common table salt (sodium chloride, NaCl), dissolve in water
very easily. The positively charged sodium ion (Na+) binds to oxygen, while the
negatively charged chloride ion (Cl-) attaches to hydrogen. This makes
a very stable “salty” solution. - This property of water allows for the transport of nutrients vital to life in animals and plants.
- A drop of rainwater falling through the air dissolves atmospheric gases. When rain reaches the earth, it affects the quality of the land, lakes and rivers.
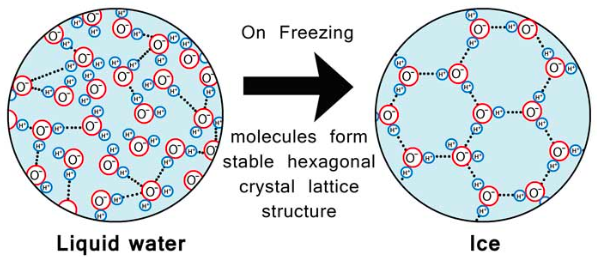
EXPANDS WHEN FROZEN
- Water is most dense at 4 degrees Celsius and then begins to expand again (becoming less dense) as the temperature decreases further.
- This expansion occurs because its hydrogen bonds become more rigid and ordered.
- As a result, frozen water (ice floats) upon the denser cold water. The expansion of water takes place even before it actually freezes.
- This explains why a pond freezes from the surface down, rather than from the bottom up.
- This ice insulates the water below from the wintry chill so that it is less likely to freeze.
- Organisms that inhabit the pond are able to survive the frigid winter below the icy surface.
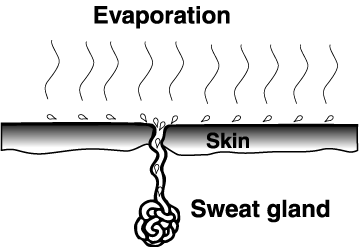
HIGH HEAT OF VAPORIZATION
- Water has a high heat of vaporization.
- Water absorbs heat as it changes from liquid to gas.
- We can dissipate excess heat by the evaporation of its sweat.
- A leaf can keep cool in the bright sunlight by evaporating water from its
surface. (Transpiration) - Water’s high heat conductivity makes possible the even distribution of heat throughout the body.
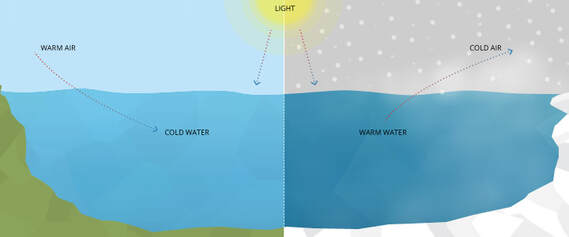
High Heat Capacity / RESISTS TEMPERATURE CHANGE
- Because so much heat loss or heat input is required to lower or raise the temperature of water, the oceans and other large bodies of water have relatively constant temperatures (MODERATION OF TEMPERATURE)
- Many organisms living in the oceans are provided with a relatively constant environmental temperature. The high water content of plants and
animals living on land helps them to maintain a relatively constant internal temperature
- Water also takes part in some metabolic reactions, for example, in
Hydrolysis and Condensation reactions. - Water also takes part in some metabolic reactions, for example, in
Hydrolysis and Condensation reactions. - Water also takes part in some metabolic reactions, for example, in
Hydrolysis and Condensation reactions.